
Sigma bonds will typically form when s orbitals from different atoms overlap to create a bond. Within a sigma bond, the electron probability clouds will be along the axis connecting the nuclei of the bonded atoms. Sigma bonds are the first type of bond that will form between atoms. When the orbitals of one atom overlap with the orbitals from another atom, they form molecular orbitals that allow for molecular bonds which, of course, allow for molecules. Electrons in orbitals farther from the nucleus will have more energy than electrons in orbitals closer to the nucleus. Orbitals are regions around atoms associated with certain energy levels. Within covalent bonds, electrons are shared and the way that they are shared is through the probability clouds of the electrons, and the orbitals in which they are located, overlapping in a manner that is roughly symmetric. In metallic bonds, electrons will be uniformly distributed through the entire molecule creating a sea of free, delocalized electrons enveloping positively charged ions attracted to the electrons. In ionic bonds, the atoms will simply exchange an electron so that one atom will become positively charged and the other negatively charged, causing them to become attracted by the electromagnetic force. Ionic bonds, metallic bonds, and covalent bonds. There are essentially three types of bonds. Molecules form when atoms exchange or share electrons through chemical bonding. It does not store any personal data.Sigma bonds are bonds between atoms within molecules formed along the axis connecting the bound nuclei of the atoms. The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. The cookie is used to store the user consent for the cookies in the category "Performance". 
This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. The cookies is used to store the user consent for the cookies in the category "Necessary". The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. One sigma bond and two pi bonds make up a triple bond. How many sigma bonds are in a triple bond? Some of the frequently asked questions are given below: 1. Overlapping orbitals perpendicular to sigma bond orbitals Overlapping orbitals perpendicular to pi bond orbitals Second bonds form between atoms within moleculesįormed from overlapping orbitals such as s orbitalsįormed from overlapping orbitals such as p orbitals The first bonds that form inside molecules are between atoms. Differences between sigma bonds and pi bonds Sigma BondĪtomic orbitals overlap along the bonding axisĪtomic orbitals overlap above and below the bonding axis They can also be stable or unstable depending on whether electrons are in bonding or anti-bonding molecular orbitals. Sigma bonds and pi bonds are both based on certain molecular orbitals that are produced from the overlapping of specific atomic orbitals, such as s orbitals for sigma bonds and p orbitals for pi bonds. Similarities between sigma bonds and pi bonds 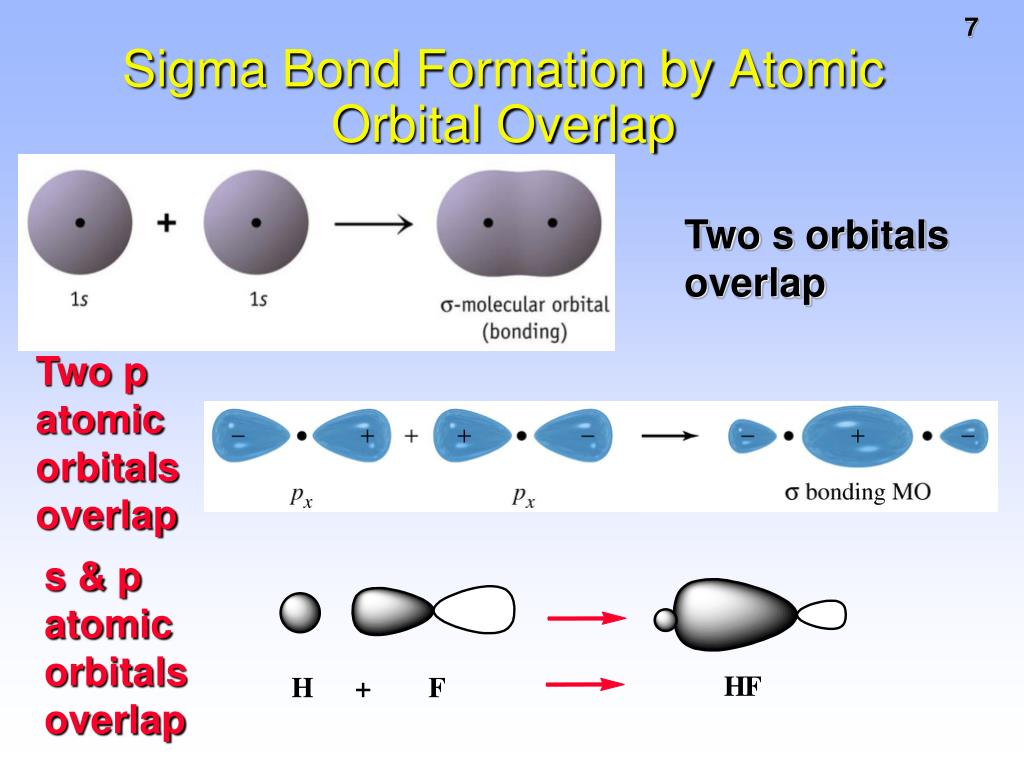
One sigma bond and one Pi bond combine to form a double bond. There are two double bonds in carbon dioxide. As a result, the sigma ( σ ) bond is more powerful than the Pi ( π) bond. The lateral overlap of the atomic orbitals that are aligned perpendicular to the internuclear axis forms a pi bond, thus the extent of orbital overlapping in a sideways manner is smaller. The pi bond is a covalent connection created by the lateral overlapping of half-filled atomic orbitals of atoms. The sigma bond is a covalent link produced by the head-on overlapping atomic orbitals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
